Fused in sarcoma undergoes cold denaturation: Implications on phase separation
Por um escritor misterioso
Last updated 20 setembro 2024

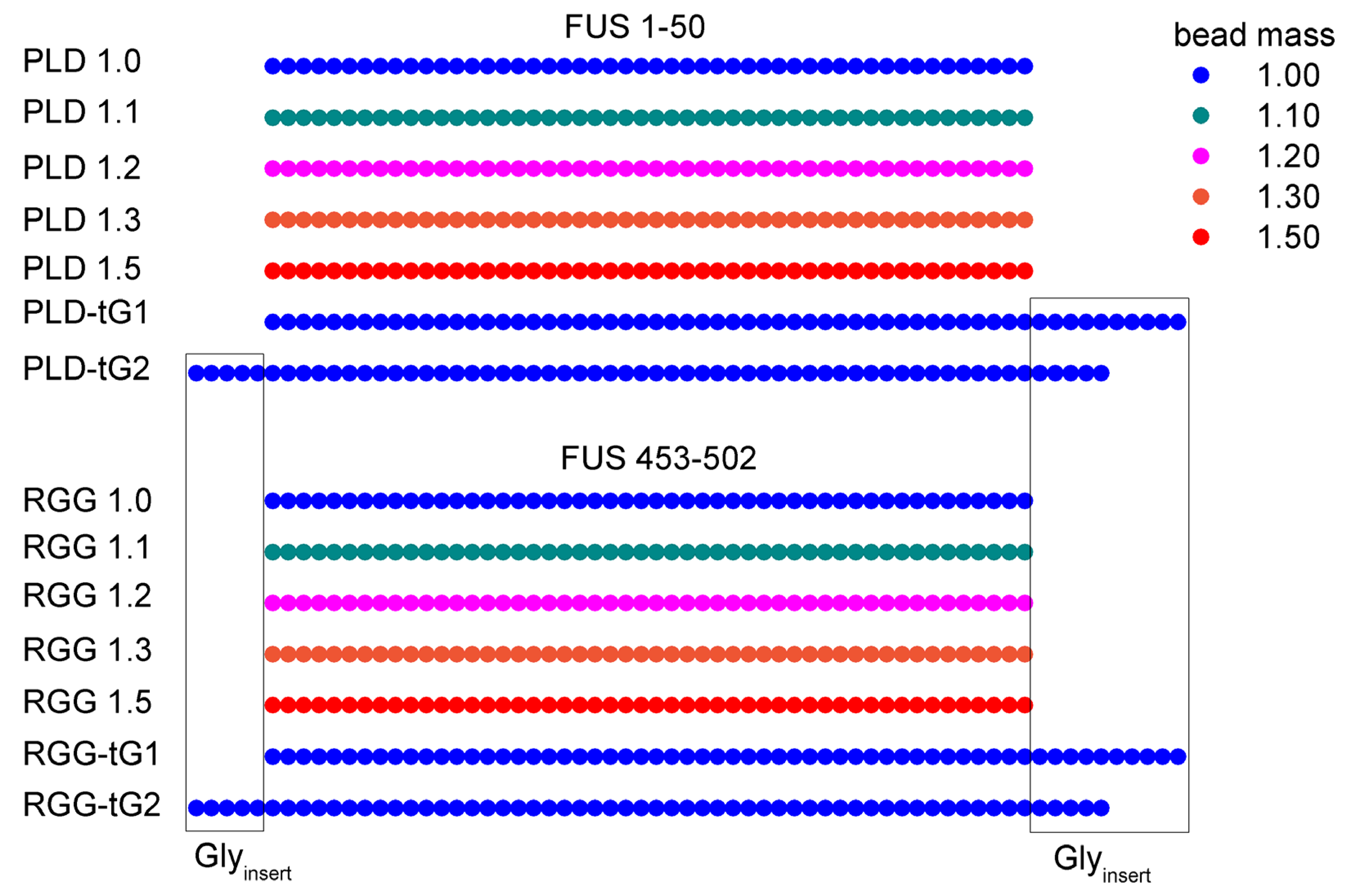
The mediation of fused in sarcoma (FUS) protein liquid-liquid phase separation (LLPS) is generally attributed to the low-complexity and disordered domains, while the role of its folded domains remains unknown. In this work we questioned the role of the folded domains on the full-length (FL) FUS LLPS and studied the influence of several metabolites, ions and overall conditions on the LLPS process using turbidity assays, differential interference contrast microscopy and nuclear magnetic resonance spectroscopy. We demonstrate that FL FUS LLPS is highly responsive to the surrounding conditions, and that overall intrinsic disorder is crucial for LLPS. To promote such disorder, we reveal that the FUS RNA-recognition domain (RRM) and the zinc-finger motif (ZnF) undergo cold denaturation above 0ºC, at a temperature that is determined by the conformational stability of the ZnF domain. We hypothesize that, in cold shock conditions, cold denaturation might provide a pathway that exposes additional residues to promote FUS self-assembly. Such findings mark the first evidence that FUS globular domains may have an active role in stress granule formation in cold stress.
Biomolecules, Free Full-Text

Promotion of Liquid–Liquid Phase Separation by G-Quadruplex DNA and RNA

PDF] Fused in Sarcoma: Properties, Self-Assembly and Correlation with Neurodegenerative Diseases

Plot of the characteristic length determined by USAXS as a function of

PDF] Fused in Sarcoma: Properties, Self-Assembly and Correlation with Neurodegenerative Diseases

The cold denaturation of IscU highlights structure-function dualism in marginally stable proteins. - Abstract - Europe PMC

Temperature-Controlled Liquid–Liquid Phase Separation of Disordered Proteins

Cold shock causes energy depletion and AMPK activation. (A) COS7 cells

Mass Spectrometry of RNA-Binding Proteins during Liquid–Liquid Phase Separation Reveals Distinct Assembly Mechanisms and Droplet Architectures

It's not just a phase: function and characteristics of RNA-binding proteins in phase separation

Biomolecular Condensates: Sequence Determinants of Phase Separation, Microstructural Organization, Enzymatic Activity, and Material Properties

with 1 supplement: 640 Download Scientific Diagram

Liquid–Liquid Phase Separation and Its Mechanistic Role in Pathological Protein Aggregation - ScienceDirect
Figure S3. Comparison of the 15 N HSQC NMR spectra of wild type Yfh1

Cold Denaturation of the HIV-1 Protease Monomer
Recomendado para você
você pode gostar











![HGJTF Phone Case for Xiaomi 12 Lite (6.55), 2 Pcs Shockproof Soft Silicone Bumper Shell, [Ultra-Thin ] [Anti-Yellowing] Clear Back Cover for Xiaomi](https://m.media-amazon.com/images/I/511kst809EL._AC_UF894,1000_QL80_.jpg)



