ANANDA Scientific Announces FDA approval of the IND for the Clinical Trial on the Treatment of Opioid Use Disorder (OUD)
Por um escritor misterioso
Last updated 19 outubro 2024

ANANDA Scientific Inc., (a biotech pharma company) today announced approval by the U.S. Food and Drug Administration (FDA) of the Investigational New

Neurological Archives - Page 2 of 21 - Drug Delivery Business

Ketamine, Esketamine, and A New Generation of Antidepressants

ANANDA Scientific Announces FDA approval of the IND for the Clinical Trial on the Treatment of Opioid Use Disorder (OUD)

Sohail R. Zaidi posted on LinkedIn

The Opioid Crisis and Recent Federal Policy Responses
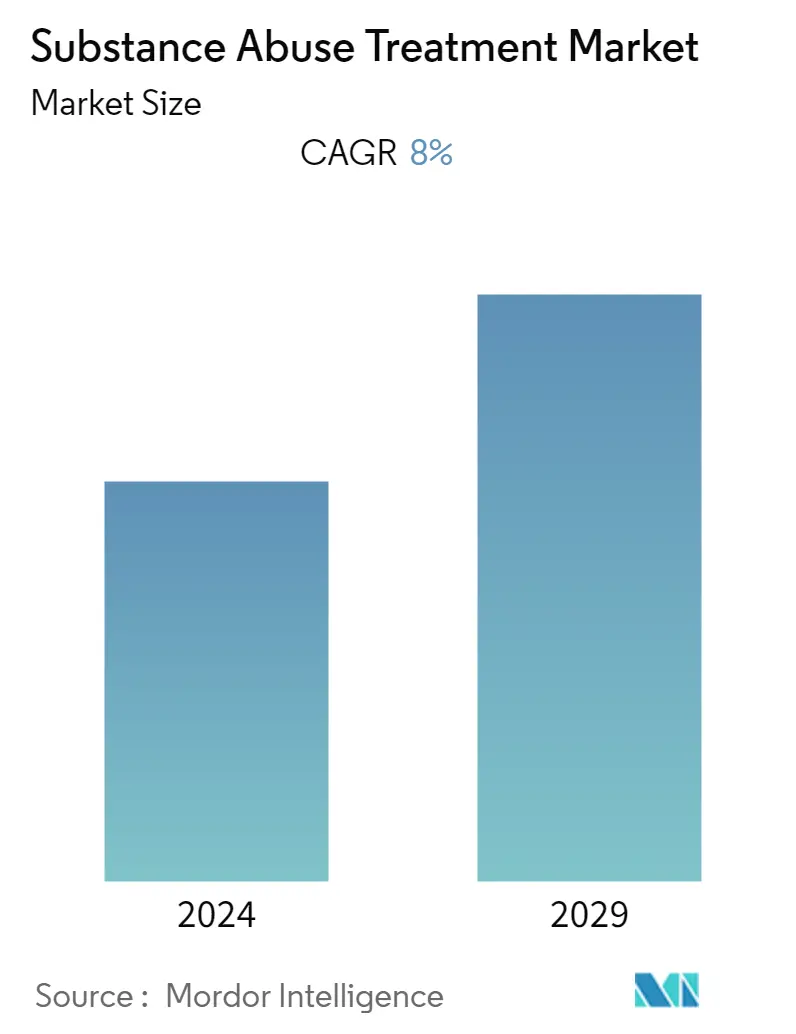
Substance Abuse Treatment Market Size & Share Analysis - Industry Research Report - Growth Trends

A Decade of FDA-Approved Drugs (2010–2019): Trends and Future Directions

Ananda Scientific (@AnandaScience) / X

Emerging mechanisms and treatments for depression beyond SSRIs and SNRIs - ScienceDirect
Recomendado para você
você pode gostar



















